Difference between revisions of "Quell relief"
(→Usage: update) |
(→Health Risks: update) |
||
| Line 142: | Line 142: | ||
<!-- Any related health issues, be it already discovered and covered in literature, or just speculative ones, should be described and properly cited in this section. --> | <!-- Any related health issues, be it already discovered and covered in literature, or just speculative ones, should be described and properly cited in this section. --> | ||
Official statement about side effects is that there are none[https://www.quellrelief.com/clinicians]. Usage is 100% drug free, with clearance by FDA (Food and Drug Administration) received in 2014. | Official statement about side effects is that there are none[https://www.quellrelief.com/clinicians]. Usage is 100% drug free, with clearance by FDA (Food and Drug Administration) received in 2014. | ||
| + | |||
| + | |||
| + | === Contraindication === | ||
== Enhancement/Therapy/Treatment == | == Enhancement/Therapy/Treatment == | ||
Revision as of 08:21, 10 November 2015
Quell is wearable device developed in purpose to provide paliative treatment for people suffering from chronical pain. Quell is commercially available, so customers don't need any prescription from a doctor.
| QUELL RELIEF | |
|---|---|

|
|
| Category | Therapeutic wearables |
| Developer | NeuroMetrix |
| Announced | Oct 2015 |
| Released | Consumers: 2015 |
| Price | 249 USD |
| Weight | 62 g |
| Dimensions | 98 x 74 x 11 mm |
| Controls | smartphone |
| Standalone[1] | |
| https://www.quellrelief.com/product | |
Contents
Main characteristics
Wearable kit with accessories
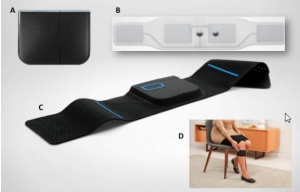
The basic product you can buy, called Quell wearable pain relief - starter kit[2], includes one piece of Quell device, adjustable sports band, charger cable, and charger adapter, and two 2 electrodes (company claims one month supply).
User have to buy accessories in addition like travel bag, spare cables, charger adapter, sports band[3]. And also two electrodes, user has to buy regularly, and which are currently available in two kinds of packages: one package of two electrodes for 29.95$, or three packages of two electrodes for 89.85$[4].
Usage
Regardless which part of body is in pain, Quell is always placed on upper calf (no matter which side). The reason is that there is better contact with nerves, on which are impulses sent to medulla. After the devices is fastened around the calf, user needs only to press the button on it.
For easier control it is possible to download Quell application for iOS[6] or Android[7].
Principle
Purpose
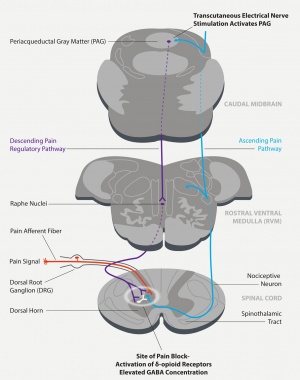
Quell wearable is device designated for treatment of chronic pain, nerve deseases and disrupted sleep[9]. It works on principle of transcutaneous electrical nerve stimulation, which causes release of endogenous opiates affecting pain relief.
Company & People
Neurometrix is company directed on developing health-care wearable technology with main effort on sleep disorders, chronical pain, and nerve deseases treatment. The company, having a seat in Waltham Massachusetts, was founded in 1996 as a spinoff of the Harvard-MIT partition of Health Sciences and Technology.[10][11]
Currently Neurometrix offers four products in total[12]:
Quell™ - Wearable device for paliative purpose
SENSUS® - Therapy device available on prescription. It is based on TENS technology also for pain relief without any drug usage.
DPNCheck® - Nerve conduction test for evaluating systemic neuropathies such as diabetic peripheral neuropathy (DPN), its detection and monitoring.
ADVANCE™ System
Directors of Neurometrix[13]:
Shai N. Gozani, M.D., Ph.D.
Allen Hinkle, M.D.
David E. Goodman, M.D
Timothy R. Surgenor
Nancy E. Katz
David Van Avermaete
Neurometrix stated also contact persons for media, which is Laura Wagstaff[14], and Thomas T. Higgins as contact person for investor relations[15].
Important Dates
1965 - Melzack and Wall proposed first conceptual model for mechanism which could lead to pain relief
1970s - Developement of TENS (Transcutaneous electrical nerve stimulation)
Ethical Issues
Health Risks
Official statement about side effects is that there are none[1]. Usage is 100% drug free, with clearance by FDA (Food and Drug Administration) received in 2014.
Contraindication
Enhancement/Therapy/Treatment
Quell wearable device has primarily therapeutical function. It was developed on purpose to palliate chronical pain which basically has origin in these four diseases:
Public & Media Impact and Presentation
Advertising video found right on the Quell product website.
Public Policy
Quell received clearance from Food and Drug Administration, which is federal agency as part of Department of Health and Human Services. FDA is connected to the database on U.S. Government Publishing Office, Electronic Code of Federal Regulations (eCFR). This eCFR contains also of three chapters (I. Food and Drug Administration, Department of Healt and Human Services; II. Drug Enforcement Administration, Department of Justice; III. Office of National Drug Control Policy), where medical devices are sorted and classified according to specific properties, difficulties of usage, dangerousness, etc. It the chapter I, Quell description consists of a) identification and b) classification. Quell is kept under regulation number: 882.5890, and it is identified as "A transcutaneous electrical nerve stimulator for pain relief is a device used to apply an electrical current to electrodes on a patient's skin to treat pain"[16] . Further, it is classified with a "Class II (performance standards)"[17]. The latter category features most of medical devices (FDA stated 43%), including for example wheelchairs of pregnancy test kits[18].
Related Technologies, Project or Scientific Research
References
- ↑ Shows if the device is a standalone wearable computer or if it needs to be connected to a processing unit to function.
- ↑ Quell wearable pain relief, starter kit, online shop webpage: https://store.quellrelief.com/products/quell-wearable-pain-relief
- ↑ Quell, Accessories, Online shop webpage: https://store.quellrelief.com/collections/accessories
- ↑ Quell, Electrodes, Online shop webpage: https://store.quellrelief.com/collections/electrodes
- ↑ Shai N. Gozani, Science Behind Quell™ Wearable Pain Relief Technology for Treatment of Chronic Pain: https://www.quellrelief.com/files/science-behind-quell.pdf
- ↑ Quell relief application available to download: https://itunes.apple.com/app/quell-relief/id972079954
- ↑ Quell relief application available to download on GooglePlay: https://play.google.com/store/apps/details?id=com.neurometrix.quell
- ↑ Introducing Quell, a 100% drug free technology proven to fight pain: https://www.quellrelief.com/files/Quell%20HCP%201%20page%20(3).pdf
- ↑ Neurometrix, Company overview: http://www.neurometrix.com/about-neurometrix/company-overview.html
- ↑ Neurometrix, Company overview: http://www.neurometrix.com/about-neurometrix/company-overview.html
- ↑ NeuroMetrix Launches Quell™ Wearable Pain Relief Technology: http://www.businesswire.com/news/home/20150615005489/en/
- ↑ Neurometrix, Company overview: http://www.neurometrix.com/about-neurometrix/company-overview.html
- ↑ Neurometrix, Board of directors: http://www.neurometrix.com/about-neurometrix/board-of-directors.html
- ↑ NeuroMetrix Launches Quell™ Wearable Pain Relief Technology: http://www.businesswire.com/news/home/20150615005489/en/
- ↑ NeuroMetrix Launches Quell™ Wearable Pain Relief Technology: http://www.businesswire.com/news/home/20150615005489/en/
- ↑ Code of Federal Regulations, Title 21, Volume 8, 21CFR882.5890 http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRsearch.cfm?FR=882.5890
- ↑ Code of Federal Regulations, Title 21, Volume 8, 21CFR882.5890 http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRsearch.cfm?FR=882.5890
- ↑ Medical device classification list on: http://www.fda.gov/MedicalDevices/ResourcesforYou/Consumers/ucm142523.htm